Introduction
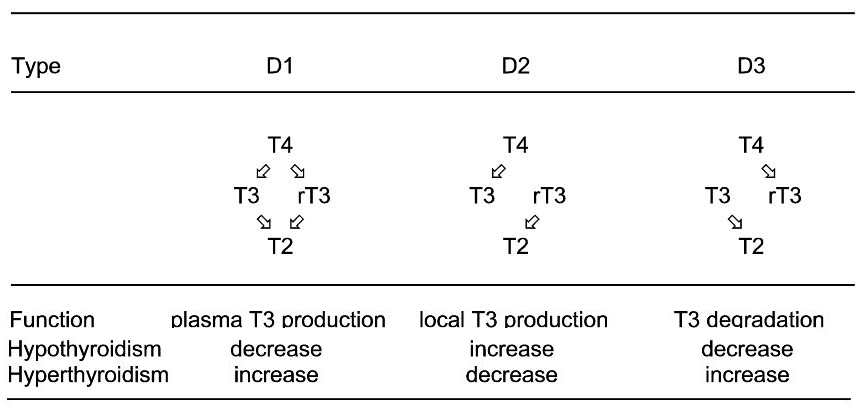
The thyroid gland secretes two major hormones thyroxine (T4) and triiodothyronine (T3). T4 is a prohormone with little hormonal activity, it needs to be converted to the active hormone T3. There is a lot more T4 than T3, T4 acts as a reservoir of hormone which is converted to T3 as and when needed. Conversion from T4 to T3 is carried out by deiodinases. There are three deiodinases: D1, D2 and D3.
T4 is converted to T3 and to ‘reverse T3’ (rT3). rT3 is often described as inactive because it does not bind to thyroid hormone receptors and consequently does not act as a thyroid hormone. T3 and rT3 have the same chemical formula but different structures, they are isomers. You can see this in the diagram below.
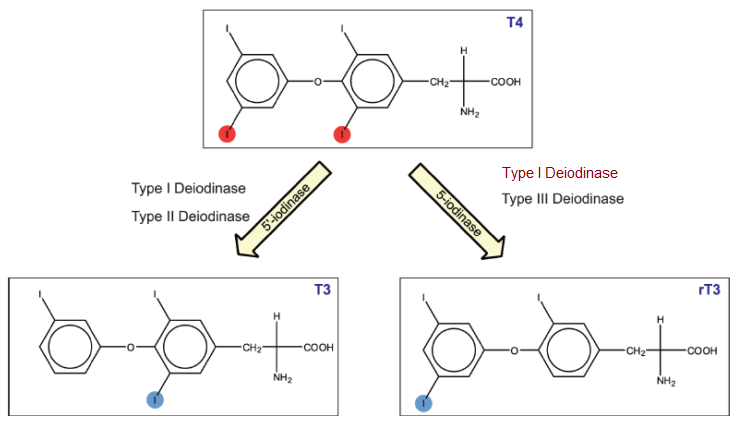
Type-1 deiodinase (D1) activity increases when T3 levels are high, it converts T4 to T3 and rT3. D1 also converts T3 to T2 and rT3 to T2. However, rT3 is very much its ‘preferred substrate’ which means D1 primarily converts rT3 to T2.
Type-2 deiodinase (D2) activity increases when T4 levels are low, it converts T4 to T3 and to a lesser degree rT3 to T2. D2 is expressed in the endoplasmic reticulum which is close to the cell nucleus in tissues such as the brain, heart, skeletal muscles and brown fat (BAT). D2 has a role in maintaining local T3 levels as well as serum T3 levels. When we run a blood test the results do not tell us how much hormone is in these D2 regulated tissues.
Type-3 deiodinase (D3) also regulates local T3 levels by converting T4 to rT3 and T3 to T2. T4 is the preferred substrate of D3, most rT3 comes from D3 action on T4.
For more information about deiodinases and why D2 and D3 are important see the topic Where is the T3 Coming From? which contains a link to the full version of the above diagram.
We will see that as well as being ‘inactive’ (in terms of thyroid hormone binding) rT3 inhibits D2 activity. So, it is wrong to describe rT3 as inactive. This role is poorly understood and complicated, we will take a chronological look at some of the more important studies. These studies fall into two categories: trials in humans or animals (in vivo) and experiments in test tubes (in vitro). in vitro studies show that rT3 inhibits D2 activity whereas in vivo studies are less conclusive.
Although this topic shows that rT3 affects D2 activity I do not recommend rT3 blood tests for monitoring hypothyroidism. They are expensive, difficult to interpret and don’t reflect local rT3 levels. Some rT3 assays may be unreliable.
Reverse Triiodothyronine Does Not Alter Pituitary–Thyroid Function in Normal Subjects
In this excellent early human study from 1984 participants were given large oral doses of rT3 but there was no change in serum TSH, T3 or T4. Nor was there any change in pulse, blood pressure or weight. Thus, rT3 doses that gave much higher levels than in non-thyroidal illness (NTI) did not affect T3 levels. If you are interested in more detail it is worth reading the ‘Discussion’ section of the study.
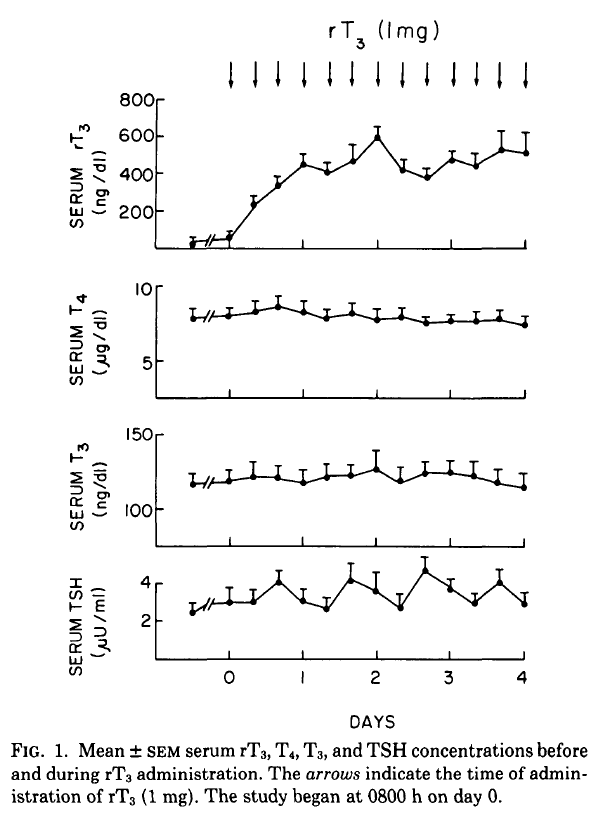
BARRY L. SHULKIN, ROBERT D. UTIGER, Reverse Triiodothyronine Does Not Alter Pituitary–Thyroid Function in Normal Subjects, The Journal of Clinical Endocrinology & Metabolism, Volume 58, Issue 6, 1 June 1984, Pages 1184–1187, https://doi.org/10.1210/jcem-58-6-1184
Regulation of Rat Cerebrocortical and Adenohypophyseal Type II 5′-Deiodinase by Thyroxine, Triiodothyronine, and Reverse Triiodothyronine
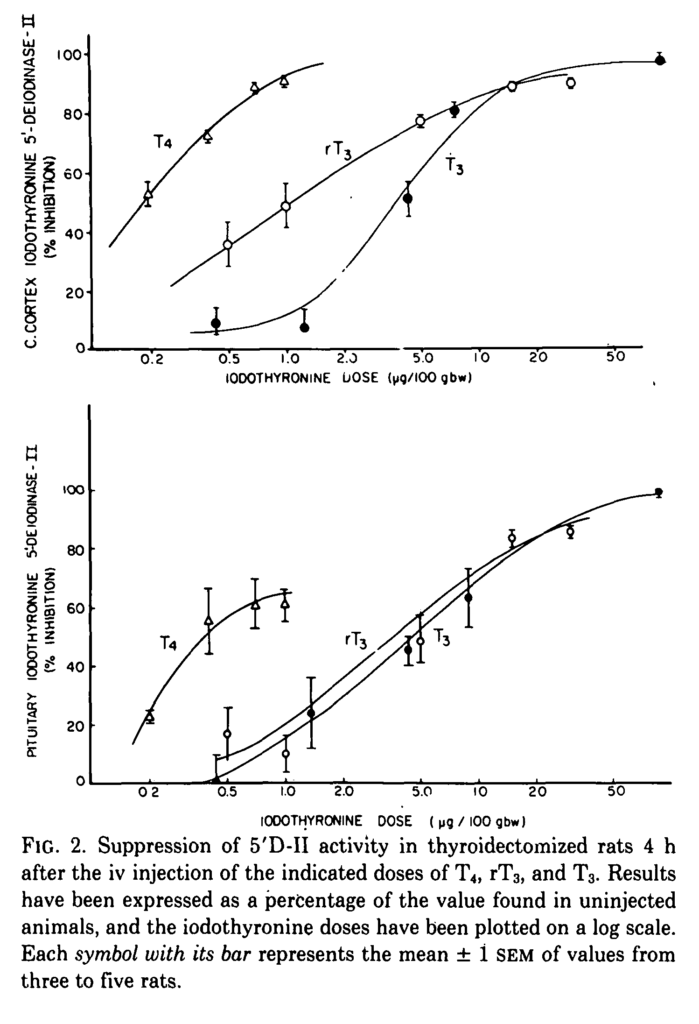
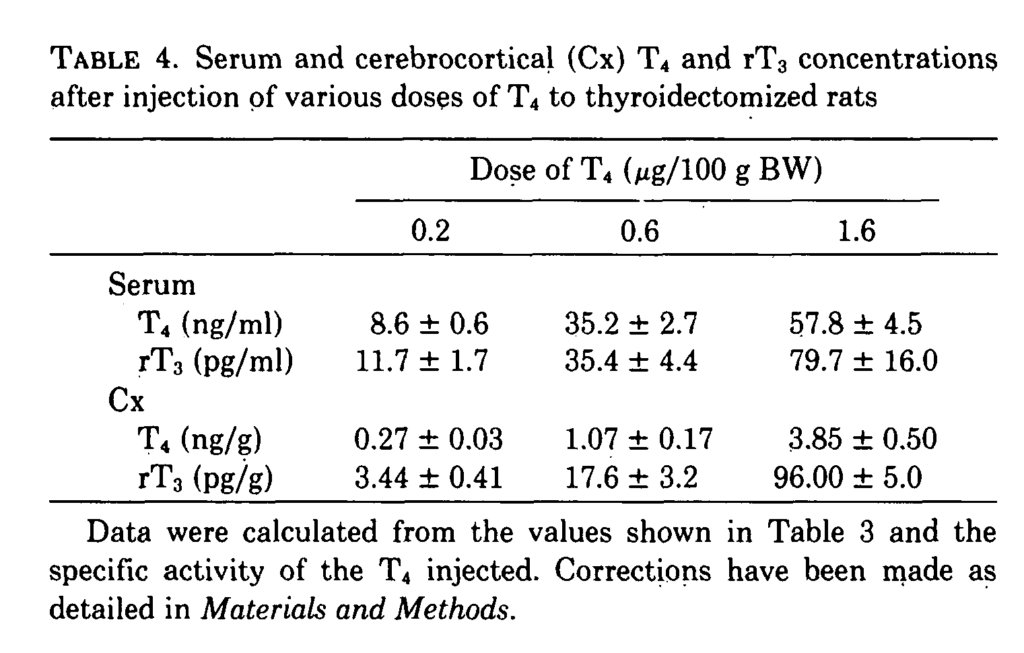
In a study from 1985 rats were injected with various doses of T4, T3 and rT3. The rate of D2 activity was examined in tissue taken from the Cerebral Cortex and Pituitary. Fig 2 below shows the extent to which D2 activity is inhibited. This study also showed that the effects on D2 were not mediated by the nuclear T3 receptor and that plasma T4 was probably the main signal regulating D2 activity.
Cerebral cortex T4 inhibited D2 to a much greater extent than rT3 which in turn was more potent than T3. rT3 inhibited D2 activity in the brain. However, this effect may be small at normal rT3 levels.
A similar effect took place in the pituitary but the potency of T3 was similar to rT3. The study also showed that TSH was substantially suppressed at low concentrations and fully suppressed at the higher concentrations. This makes sense, if T4 stopped D2 at normal concentrations the pituitary would not be able to respond to T4.
Bear in mind this study shows the effects of T4, T3 and rT3 four hours after injection. rT3 has a short half-life so these effects will have occurred at substantially reduced concentrations. Most rT3 is produced by D3, so local concentrations of rT3 might be higher.
J. ENRIQUE SILVA, JACK L. LEONARD, Regulation of Rat Cerebrocortical and Adenohypophyseal Type II 5′-Deiodinase by Thyroxine, Triiodothyronine, and Reverse Triiodothyronine, Endocrinology, Volume 116, Issue 4, 1 April 1985, Pages 1627–1635, https://doi.org/10.1210/endo-116-4-1627
The role of 3,3′,5′-triiodothyronine in the regulation of type II iodothyronine 5′-deiodinase in the rat cerebral cortex
This study follows on from the previous one. The objective was to find whether endogenous levels of rT3 regulate cortical D2. Fig. 1 shows that when rT3 was injected it suppressed D2 activity. Suppression depended on the cerebrocortical concentration of rT3 with a linear inverse relationship between D2 activity and log(rT3). At euthyroid levels of rT3 D2 was 20% to 30% suppressed.
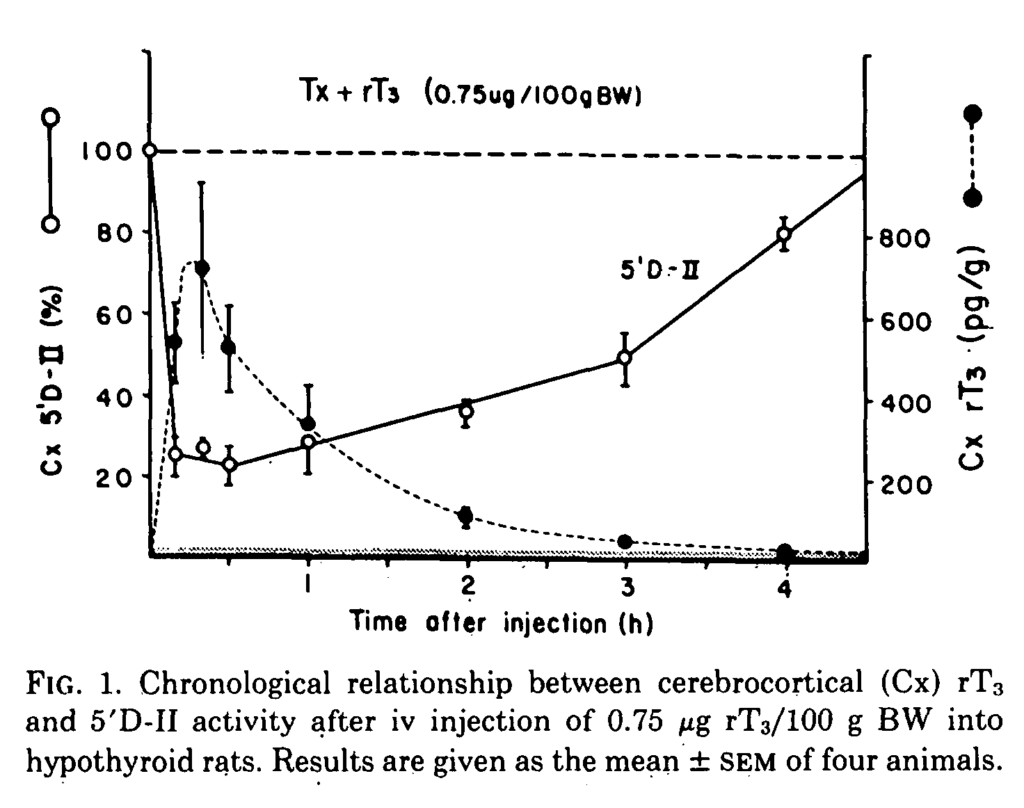
When T4 was injected both serum and cerebrocortical rT3 increased but with a much greater effect in the cortex where the rate of increase in rT3 was steeper than that of T4. The study confirms that rT3 is a very potent suppressor of D2. Plasma rT3 made a small contribution to cerebral rT3 and thus only rT3 generated in situ was likely to be physiologically relevant.
Expression of Type 2 Iodothyronine Deiodinase in Human Osteoblast Is Stimulated by Thyrotropin
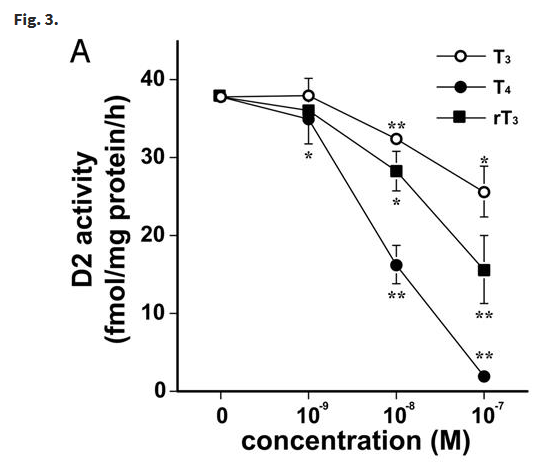
This in vitro study also found that rT3 inhibits D2 activity in osteoblasts. Very similar results were found in three other in vitro studies with rT3 inhibiting D2 activity in human skeletal muscle cells, human vascular smooth muscle cells and mouse pituitary tumour cells. These studies show that rT3 suppresses D2 activity, at least at the cell level. It remains to be established to what extent this happens in euthyroid humans.
The study found that D2 is degraded by ubiquitin and so its half-life is reduced.
Inhibition of pituitary type 2 deiodinase by reverse triiodothyronine does not alter thyroxine-induced inhibition of thyrotropin secretion in hypothyroid rats
This study infused rT3 into homogenized pituitary, brain cortex and brown adipose tissue (BAT). D2 activity was substantially reduced in all tissues including the pituitary but TSH remained unchanged. Perhaps TSH is regulated primarily by the hypothalamus. T4 and rT3 inhibit D2 activity by ubiquitination which is not upregulated by T4 in the hypothalamus. Another possibility is that homogenized tissue behaves differently to in vivo tissue. Perhaps pituitary D2 activity is retained (other than in the thyrotrope?) so that secretion of other pituitary hormones can be maintained. The evidence shows that rT3 inhibits D2 but does not affect the thyroid axis.
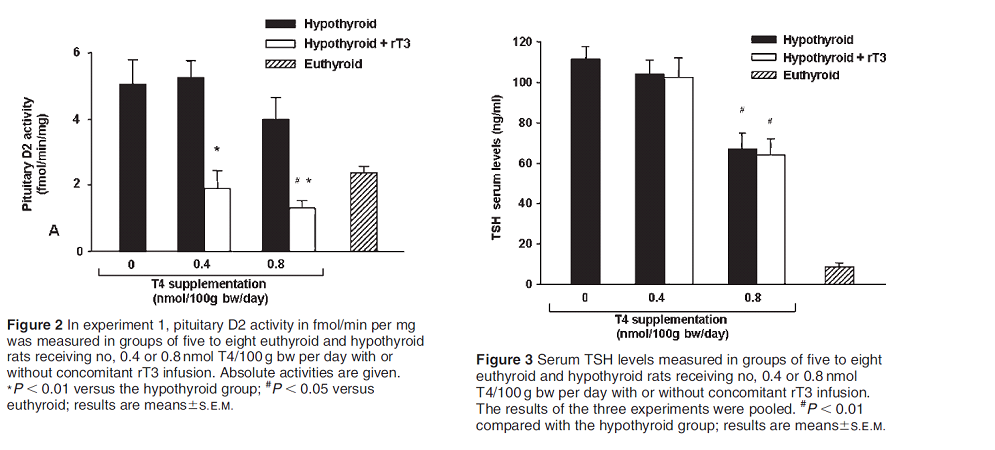
In spite of the marked inhibition of D2 activity by rT3 there was no change in serum TSH or T4 levels and only a small decrease in T3 levels. Similar results have been found in other studies.
Disruption of Type 2 Iodothyronine Deiodinase Activity in Cultured Human Glial Cells by Polybrominated Diphenyl Ethers
rT3 is now accepted as an inhibitor of D2 activity and was used as a control marker in this 2015 study that measured the effects of an environmental disrupting chemical (EDC) on D2 activity in human brain cells. This study showed that rT3 inhibits D2 activity in astrocyte cells. Astrocytes are the main source of T3 for neurons in the human brain.
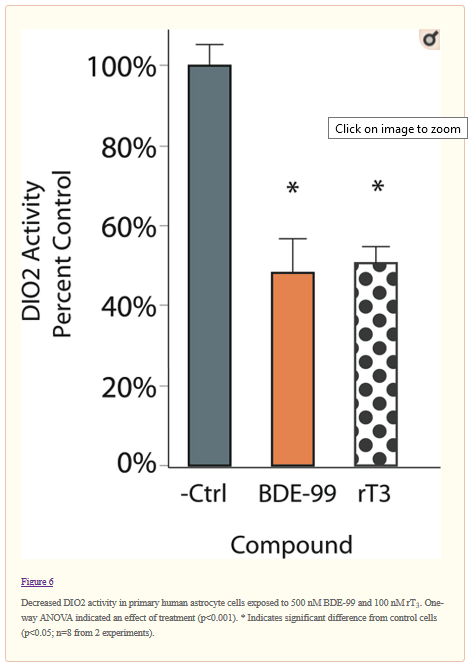
Reprinted with permission from Chem. Res. Toxicol.20152861265-1274
Publication Date: May 24, 2015
https://doi.org/10.1021/acs.ch https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4827872/emrestox.5b00072 Copyright 2015 American Chemical Society.
Reverse triiodothyronine (rT3) attenuates ischemia-reperfusion injury
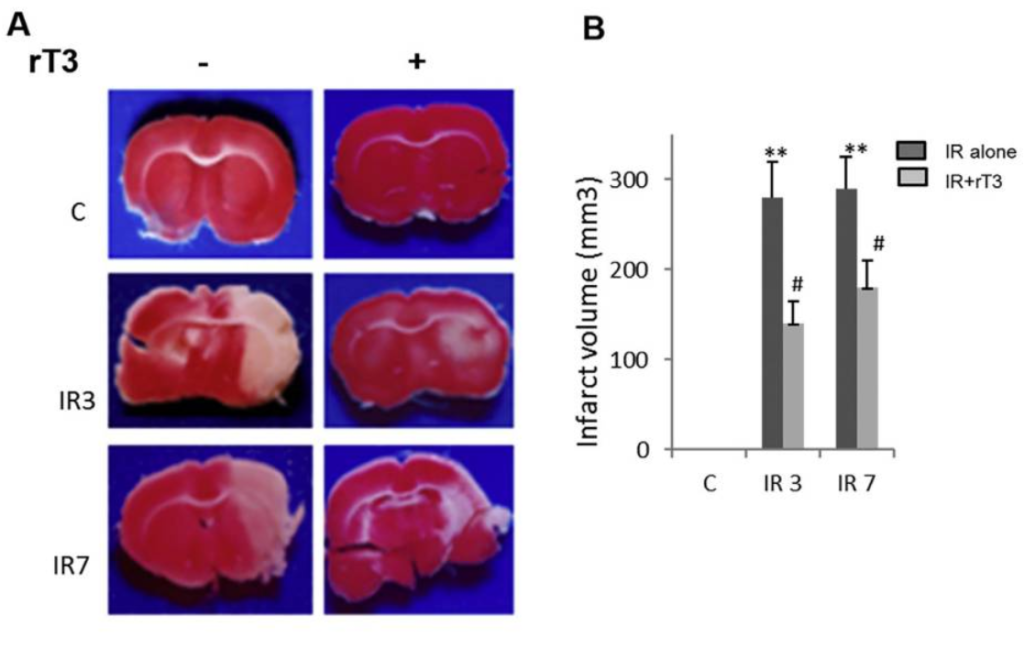
rT3 can cross the blood / brain barrier and inhibit D2 activity without altering circulating T3. Low thyroid hormone levels attenuate, and high levels aggravate ischemic damage. This experiment tested the hypothesis that rT3 can produce a brain specific hypothyroid state. i.e. rT3 can cause brain hypothyroidism without affecting other organs or the thyroid axis.
The cerebral artery was occluded followed by gentle reperfusion over the next 3 and 7 days. This causes ischemic damage. As can be seen in the image below the rats who received supplementary rT3 (50ng/100g body weight) suffered considerably less injury. This demonstrates that the rT3 reduced brain T3 levels. Furthermore, rT3 had no effect on circulating T3, T4 levels, blood pressure or body temperature. People with brain injuries are sometimes put in an induced coma, this study suggests rT3 could be used in a similar manner to induce a state of brain hypothyroidism.
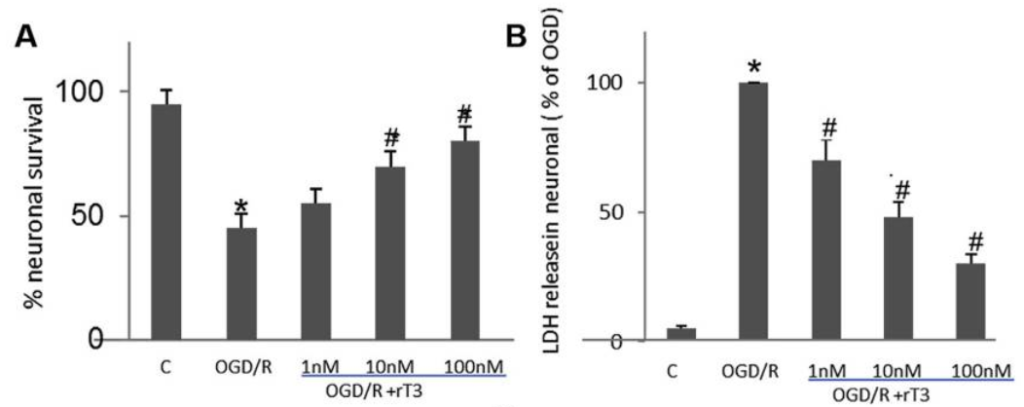
In a second in vitro experiment neurons were subjected to oxygen / glucose deprivation followed by re-oxygenation with glucose (OGD/R). The addition of rT3 was shown to protect against cell death in a dose dependant manner. (LDH is a marker for cell death).
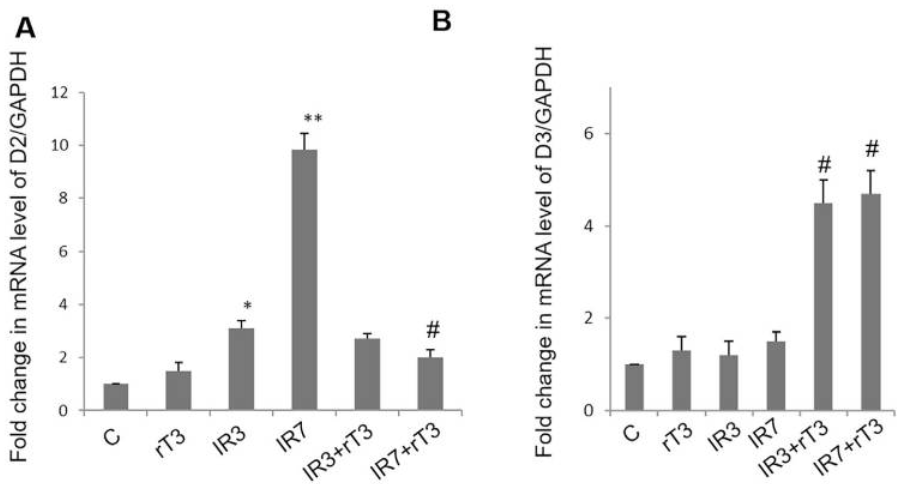
mRNA expression of D2 and D3 were measured (this tells us how much D2 and D3 is being created). The inclusion of rT3 reversed the large increase in D2 expression seen in cells without rT3 and greatly increased D3 expressioon. Thus, rT3 introduces a state of local hypothyroidism. We have seen earlier that rT3 reduces D2 activity via faster turnover caused by increased ubiquitination. This experiment shows that in brain cells it also reduces the amount of D2 created.
Summary
Studies show rT3 reduces D2 and increases D3. This is clearly demonstrated by in vitro studies but the scale of its effect in real life is not clear. Since most rT3 comes from D3 activity which takes place in organs such as the brain it is possible these organs will have local hypothyroidism when serum rT3 is high, such as during periods of starvation, severe illness or high T4 levels.
Some people have chronic high rT3 levels with no obvious cause. Studies which compare signs and symptoms in these subjects with controls could reveal the extent to which elevated rT3 reflects clinical status, if it does at all. It is difficult to assess the usefulness of rT3 levels since they vary considerably within and between individuals. Locally generated rT3 has effects on deiodinase and so in this respect rT3 is an active hormone.