We have seen how T4 acting at the integrin αvβ3 receptor promotes the growth and metastasis of cancer cells. Levothyroxine monotherapy is associated with higher fT4 levels which may lead to increased risk of cancer and mortality. Patients on levothyroxine therapy tend to have more stable hormone levels than the general population as treatment is targeted to blood test results and symptom resolution. If levothyroxine monotherapy continues to be licensed it must be as safe as any alternative therapy.
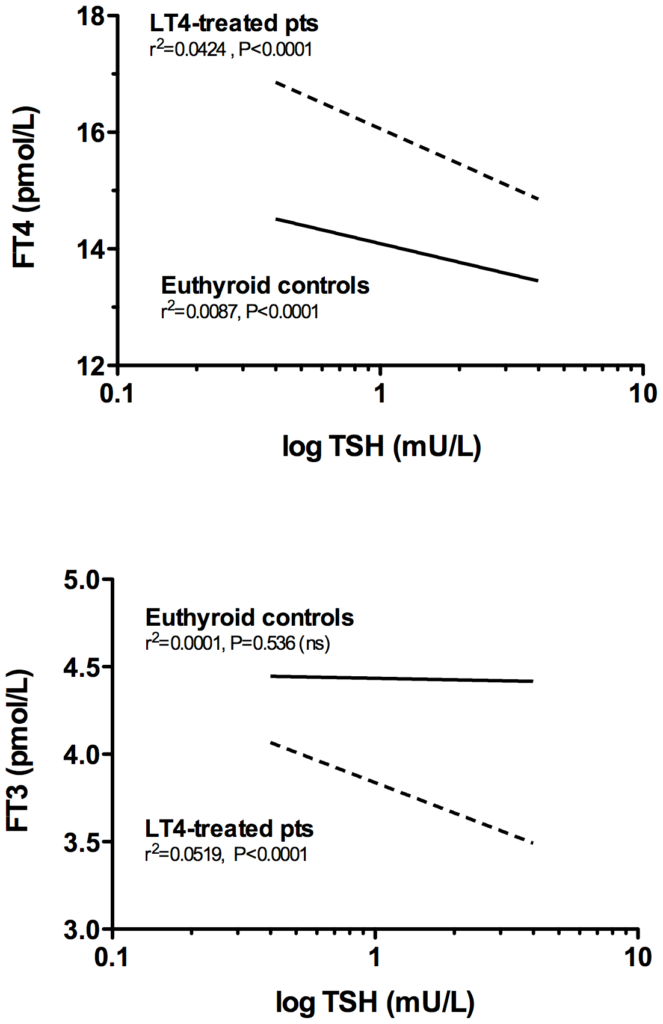
FT3 / FT4 Ratios in Levothyroxine Monotherapy and the General Population.
Several studies show that levothyroxine monotherapy lowers the fT3/fT4 ratio resulting in a higher fT4 (dotted line). This raises concern about the risk of higher cancer incidence and mortality.
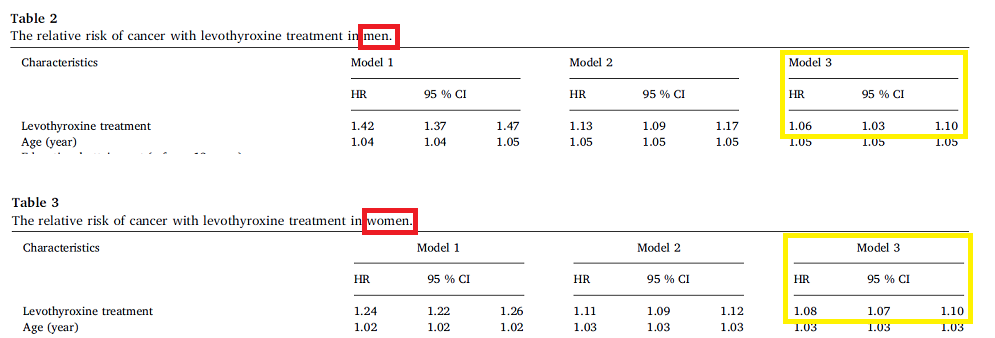
Levothyroxine is Associated with Increased Risk of Cancer – Survey of Swedish Adult Population.
This survey covered the entire adult population of Sweden (N = 8,373,313) and identified 253,193 (3%) who had received at least two prescriptions of levothyroxine. It excluded all those with cancer prior to the study or within the first two years. The results were adjusted for factors that might affect the results. Some factors were not available, such as smoking, BMI and exercise. The study found a slight increase cancer risk associated with levothyroxine therapy with HR 1.06 for men and HR 1.08 for women. Note the very narrow 95% CI ranges and all models have increased HRs.
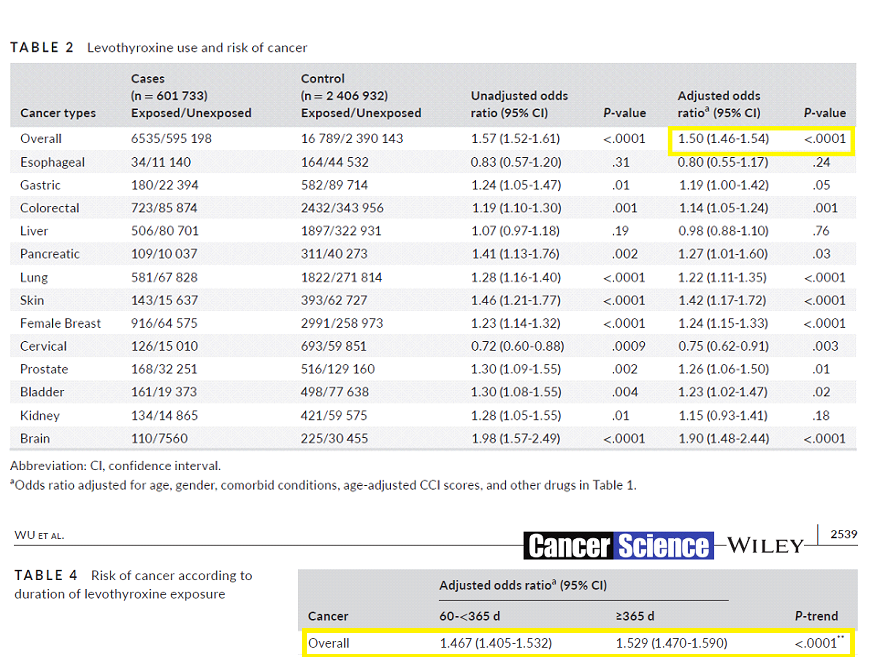
Risk of Cancer in Long-term Levothyroxine Users.
A study of cancer risk in the entire population of Taiwan found an adjusted HR of 1.5 for the levothyroxine group. Like the Sweden study it had highly significant P values and crucially the HR increased with duration of levothyroxine therapy. The authors concluded: “our study findings showed that levothyroxine use is associated with increased risk of cancer … the current study does not advise against use of this medication“.
Is Combination Therapy Safer?
Liothyronine Use in Hypothyroidism and its Effects on Cancer and Mortality,
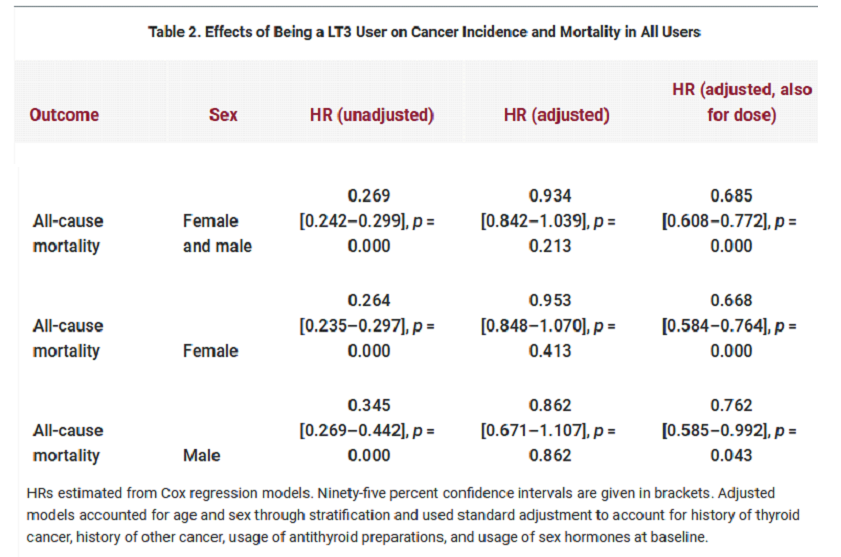
This study looked at levothyroxine and liothyronine use in the entire population of Sweden. An LT3 user was defined as someone who has ever been a user of LT3. This will include some patients who were short term or trial users of LT3 and so the results are likely to be watered down and effects underestimated. Most patients on combination therapy were previously on levothyroxine monotherapy. It’s possible that some patients with pre-existing cardiac conditions have been refused liothyronine thus biasing non-cancer mortality figures. On the other hand, many patients on combination therapy have severe symptoms for years before they are able to obtain liothyronine, a bias in the opposite direction.
When comparing cancer risks between levothyroxine monotherapy and combination therapy it is also important to consider overall mortality. From Table 2 (below) we see that dose adjusted combination therapy was associated with less all-cause mortality than levothyroxine monotherapy. Even with higher doses combination therapy had a non-significant tendency towards reduced mortality. Note the substantially reduced HRs and extraordinary low ‘p-values’.
Observe the dose adjusted effects of combination therapy on cancer risk and cancer mortality. In the special case of breast cancer there is no significant difference between monotherapy and combination therapy. Similarly, there is no difference in the risk of getting any cancer whereas two earlier studies found an increased risk. However, cancer mortality was substantially reduced in combination therapy. Patients on levothyroxine monotherapy with cancer were 29% more likely to die.
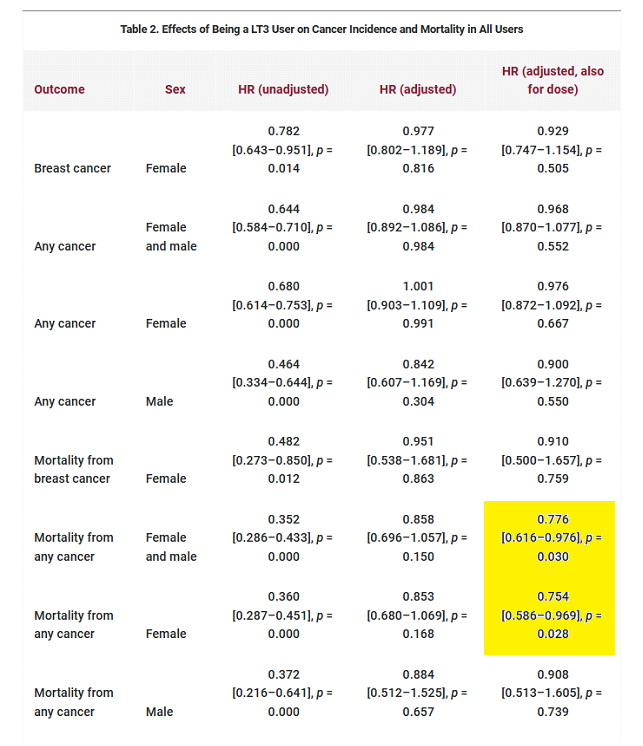
There were shorter follow-up times for patients receiving liothyronine which might have led to an underestimation of mortality from slow growing cancers. Combination therapy tended to be protective against larger cancers (‘Cancer T3/T4’) and metastasis (‘Cancer M1’). An alternative interpretation is that levothyroxine monotherapy is associated with more aggressive cancers.

Increased mortality with similar incidence strengthens the reliability of these results as selective bias is unlikely yet mortality is higher.
A Review of the Above Paper.
This review concludes: “L-T3 treatment was not related to increased cancer risks but instead had a protective effect on overall mortality as compared with LT-4 only treatment“.

“By analyzing the cases of severe cancer (i.e., cancer with more extensive spread), similar results were shown; L-T3 use was strongly protective against cancer and all-cause mortality.“
Specialists Call for T3 Treatment of Patients with Primary Hypothyroidism Who Also Have Cancer.
A chapter in the book “Hormone Therapy and Replacement in Cancer and Aging-related Diseases” recommends that T3 monotherapy is considered for patients with primary hypothyroidism who have cancer.

Summary
Levothyroxine monotherapy distorts the fT3/fT4 ratio leading to enhanced T4 action at the integrin αvβ3 receptor and a consequent increase in risk of cancer and mortality. It has been suggested that L-T3 monotherapy be considered for patients with primary hypothyroidism who have cancer.
Finally, some concluding remarks.