The enteric nervous system, sometimes called the ‘small brain’ controls the digestive system: motility, secretion, absorption of nutrients and sensory responses. We have already covered enteric sensations that are perceived as pain. Sensory perceptions of various forms (e.g. stretch, thermal, chemical) are also transmitted throughout the ENS activating responses such as peristalsis and secretion.
Abnormal Migrating Motor Complex (MMC) in IBS
The migrating motor complex consists of waves of electrical activity that occur between meals and trigger peristaltic waves. Although a very complex system, we only look at how it differs in IBS patients as observed in older studies. Research over recent decades has been biased by the presumption that IBS is ‘functional’ and primarily associated with the patient’s brain. Other research has focussed on interventions that mitigate individual symptoms – pharmaceutical companies don’t invest in finding a cure, long term intervention with expensive drugs is profitable.
The MMC has two basic electrical rhythms with frequencies of 6 cycles per second and 3 cycles per second along with spike activity. Snape discovered IBS patients have abnormal MMC activity. In normal subjects the major component was 6 cycles per minute without any spike or spontaneous contractile activity. In IBS patients the dominant frequency was 3 cycles per second accompanied by spike and contractile activity.
In spite of different myoelectric activity there was no difference in basal motor activity between patients and normal subjects. Nor was there a difference in myoelectric activity between patients with different symptoms or longer duration of symptoms.
It is suggested that symptoms of IBS are a consequence of the ‘brain gut axis’ or related to traumatic life events. Experimental observations suggest an elementary abnormality, perhaps a lower activation threshold for 3 cycle per minute activity. It’s difficult to find a psychological explanation with direct effects that preserves the waveform and frequencies, we would expect more chaos.
The authors of the study offer a possible explanation: ‘The low-frequency BER may serve as the underlying substrate upon which other factors act to elicit symptoms and to alter colonic transport. Thus, the irritable bowel syndrome may be due to a combination of certain emotional, hormonal, or neural factors acting upon a colon which has the intrinsic ability to respond to these stimuli.‘. (We address this ‘intrinsic ability‘ in the next section.)
Many studies have shown that there are abnormal patters of motor activity in IBS patients subject to stress, feeding, drugs, gastrointestinal hormones and rectal distension. In a follow-up study it was shown that the frequency of colonic contractions matched the electrical slow wave frequency, in both IBS patients and normal subjects. Although IBS patients had more 3 cycles per second slow wave activity, they had the same level of motor activity whilst in the basal state. The increase in slow waves at 3 cycles per minute was not reflected in 3 cycle per minute motor activity.
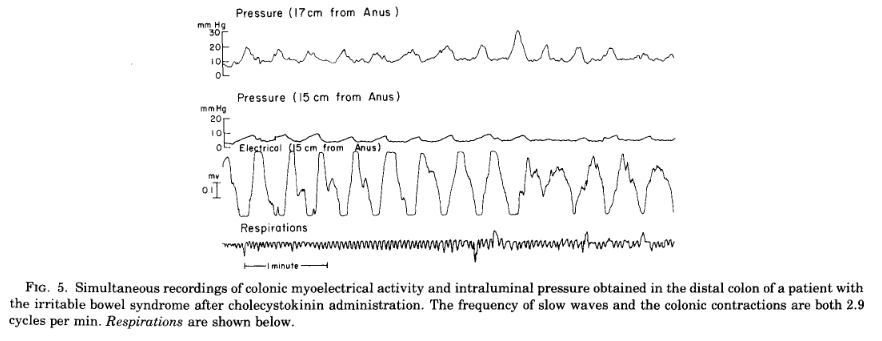
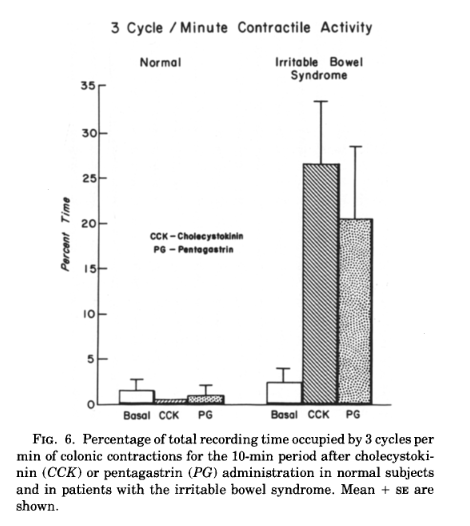
Subjects were given injections of the gut hormone cholecystokinin (CCK) or the drug pentagastrin. This made no difference to the total slow wave activity in patients or normal subjects, but spike potentials and motility increased equally in both groups. However, in the IBS group after CCK injection all 3 cycle per minute slow waves were now associated with 3 cycle per minute motor activity as can be seen in Fig. 5. There was also a large increase in 3 cycle per minute contractions (Fig. 6) in IBS patients.
These powerful slow wave contractions are associated with symptoms in IBS patients.
Chaotic MMC in IBS
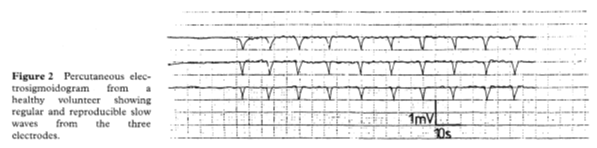
Shafik measured the electrical activity by applying three electrodes in an inverted triangle on the lower tummy. In normal subjects the slow waves were regular, with the same frequency and amplitude at each of the three sites (Fig 2).
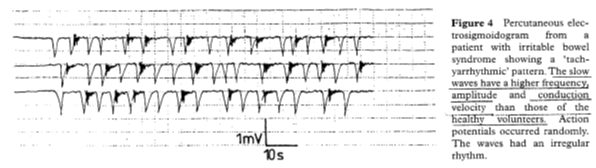
In IBS patients the slow waves had a higher frequency, greater amplitude and ‘action potentials’ (the intense dark part after the spikes). The rhythm was irregular and different at each site.
So, not only does the MMC differ in IBS patients, it crucially varies by location. If IBS symptoms were a consequence of a brain abnormality, we would expect a similar pattern at each location.
NMDA Receptors and the Enteric Nervous System
NMDA receptors not only carry pain sensations to the brain, they also carry signals within the Enteric Nervous System (ENS). In a general NMDA receptor disorder these signals would be disrupted. A gut with a lower threshold for contraction receiving sensations from and sending messages via abnormal NMDA receptors would produce the pain and chaotic contractions experienced by IBS patients.
To understand the origin of visceral sensitivity and abnormal NMDA receptor response we must investigate Magnesium Deficiency.